
 |
|
DECADES-OLD AMATEUR SCIENCE RESEARCH
STARRING OR MENTIONING DONALD RUMSFELD
From the Pipe Up! Archives
S. Manning
Chem Study-6 Pettus
May 18, 1985
Aspartame: Sweet or Sour?
The sweetening agent Aspartame was discovered in 1966, when a scientist working on an ulcer drug absentmindedly licked his fingers and discovered that the powdery substance had a sweet taste. 1 This spontaneous discovery reflects the subsequent “absentminded” methods of testing done in the efforts to approve aspartame for public use.
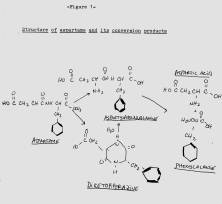
The stability of aspartame has been a question since its discovery. Aspartame is a dipeptide of L-aspartic acid and L-phenylalanine methyl ether. (See figure below). In moisture of any sort the ester and amide (peptide) bonds are subject to hydrolysis. 2 The ester bond goes first. In aspartame, the alcohol is replaced by the amino acid group of the aspartic acid, resulting in the cyclic diketopiperazine (DKP). The dipeptide aspartylphenylalanine can be produced by either hydrolytic opening of the DKP ring or directly by hydrolysis of the methyl ester. The dipeptide can ultimately hydrolyze to the individual amino acids. None of these products are sweet, but there is no bad taste either. 3 A variety of tests have been done to establish the longest lasting sweetness; tests ironically more useful than were done to test the safety of the chemical before its approval by the Food and Drug Administration.
The FDA first approved aspartame in 1974 as a sweetener and flavor enhancer for table use, and for use in certain dry food applications. Many questions arose concerning the toxic effects of certain amino acids found in the breakdown products. The release to the public was postponed for the time. A Public Board of Inquiry was appointed to research the testing submitted by G.D. Searle (manufacturer of aspartame under the NutraSweet label). After six years of research and a Grand Jury investigation, the Board recommended that it not be used for it could cause brain tumors. The Grand Jury investigation had been requested by the FDA General Counsel, Richard Merrill, when the Board found serious flaws in the tests submitted. 4 Ninety of the 113 tests had been done in the early to mid-70s, and the Board concluded that there were very serious deficiencies in operations in testing practices from 1967 to 1975 in Searle and its testing subsidiary Hazleton Laboratories. As the Grand Jury investigation of the testing continues, many scientists are researching the testing that was done.
Dr. Richard Wurtman of MIT found in a rat study that an aspartame diet, especially with carbohydrates, caused changes in brain chemistry. The production of serotonin was reduced. Normally production increases with sweet consumption, satisfying the craving for sweets. This raises the question of whether a diet drink will actually help you lose weight. 5 The lack of serotonin also causes a desire for more carbohydrates and more aspartame, which brings aspartame to high levels, a concern many scientists have. 6
One such scientist is Dr. William Pardridge of UCLA, who questions the FDA’s appointment of the maximum daily intake as 10 milligrams per kilogram. He claims that a 5-year old could easily ingest 31mg by his afternoon snack. He is also one that believes that children are more susceptible to the effects of aspartame.
Dr. Harvey Levy of Children’s Hospital in Boston works with patients with phenylketonuria (PKU-a hereditary disease). These children lack an enzyme that breaks down the phenylalanine and can become severely mentally retarded if untreated. This brings up the question of whether pregnant women should consume aspartame. Supporting Levy’s hypothesis, Dr. John Olney of Washington University conducted experiments in which infant mice developed brain abnormalities when fed aspartame. 7
Even in the tests submitted by Searle, 13 rats fed aspartame developed tumors as opposed to 1 in the control group. In another on monkeys by Searle, the monkeys exhibited seizure activity and contracted Shigella infection. One monkey died after being fed aspartame for 300 days. The Bressler Report investigation of the tests in 1972 found that one Searle scientist submitted a written evaluation of a tissue mass, supposedly found in one of the rats. They were non-cancerous polyps, but polyps nonetheless. They could not find the bottle later though, but the incident displays the disorganization of the testing. 8In the human study, those taking aspartame complained of constipation, increased menstrual cramps, headaches, increased menstrual flow, tiredness, and general swelling. There were few complaints among the placebo-takers. Finally, the insulin study of 1957 diabetics aged 21 to 70 lasted 13 weeks. In the 11th week, two women underwent cancer operation, one in the breast, the other in the stomach. 9These are the tests relied on by the FDA for their approval, for the others were non-conclusive, and even these tests were never repeated.
Another concern is that aspartame affects brain chemistry and therefore behavior. The FDA received 600 consumer complaints since the approval in 1983 for use in soft drinks. 10 People complain of headaches, rashes, dizziness, menstrual problems, and seizures. 11 One lady stated in court that her 5-year old son had suffered adverse reactions to aspartame including headaches, uncontrollable behavior and slurred speech. Others complain of numbness, insomnia, and nausea. 12
This serious situation has become quite a political comedy. Reagan appointed Arthur Hull Hayes FDA commissioner and within three months in office Hayes gave the approval of aspartame despite the serious unresolved questions surrounding the validity of the tests used in his decision. Then he approved the use in soft drinks in 1983, just before he left to go work for Searle’s PR firm.G.D. Searle has hired Republican Donald Rumsfeld to lead the firm because Searle wanted “someone with a boy scout image.” 13 The National Soft Drink Association drafted a formal objection to the approval of aspartame for soft drinks without further testing, but now the dollar signs have clouded their eyes. 14
The question remains as to whether aspartame can cause cancerous brain tumors, whether it can affect brain chemistry and therefore behavior, and the potential adverse effects on children and pregnant women. The questions on quantity and compatibility remain, as in Wurtman’s theory that the effects are worsened by the addition of carbohydrates, and Olney’s belief that consumption with monosodium glutamate increases chances of cancer. The fight continues with various interest groups’ efforts to remove the product from the market completely, or at least until further testing is done. The bad thing is that the burden of proof lies upon the people to prove aspartame is unsafe, when it should be the manufacturer’s responsibility to prove to the FDA that their product is safe within “reasonable certainty.” G.D. Searle never did prove that aspartame is safe within any certainty, but the FDA approved it anyway.
So it goes.
1 Florence Graves,
“How Safe is Your Soft Drink?” Common Cause, 10 (July
1984) p. 28.
2 Aspartame Stability,” Food Engineering, 56 (May 1984) pp.
127.
3 Ibid., p. 128
4 Florence Graves, “How Safe is Your Soft Drink?” Common Cause,
10 (July 1984) p. 28.
5 Graves, Florence. “But Will You Lose Weight?” Common Cause,
10 (July 1984) p. 42.
6“Aspartame: Too Good to be True?” Food Engineering,
56 (March 1985) p.16.
7 Ibid p. 17.
8 Florence Graves, “How Safe is Your Soft Drink?” Common Cause,
10 (July 1984) p. 28.
9“Aspartame: Too Good to be True?” Food Engineering,
56 (March 1985) p.17
10“Food Additives Permitted for Direct Addition to Food for Consumption:
Aspartame”
FDA Federal Register, 48 (1983) p. 31376
11 Graves, p. 25
12 Graves, Florence, “Does NutraSweet Cause Adverse Reac-tions?”
Common Cause 10 (July 1984) p.36
13 Florence Graves, “How Safe is Your Soft Drink?” Common
Cause, 10 (July 1984) p. 28.
14 Ibid p. 28
Please submit topical material from the past for our new feature Piping Up Through the Ages.
© 2003 Little Commie LLC About Email Message Board